Oxandrolone
-
CAS
53-39-4 - Formula C19H30O3
- Half-life 9 hours (oral)
- Detection 3 weeks
Clinical Notes
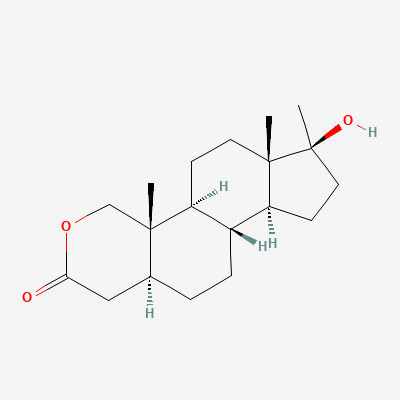
DHT-derived oral with an oxygen atom substituting C-2 on the A-ring — a modification that simultaneously protects the molecule from hepatic metabolism and neutralises 5alpha-reductase affinity. Clinical FDA indications (HIV wasting, severe burn recovery, Turner syndrome) were approved on the strength of its anabolic-to-androgenic dissociation, which is the widest in the oral AAS class. Aromatisation is mechanistically impossible. Hepatotoxicity is real but mildest of C17alpha-alkylated compounds — ALT typically rises 1.5–2.5x ULN at 40 mg/day x 8 weeks. Lipid profile degradation is severe — HDL suppression 50%+ is reproducible. Effect at 20–40 mg/day: strength gain without water retention, minimal scale weight change (5–8 kg lean mass maximum), visible muscle hardening.
Chemical Identity
| CAS Number |
53-39-4 |
|---|---|
| IUPAC Name | (1S,2S,7S,10R,11S,14S,15S)-2,15-dimethyl-5-oxotetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl acetate |
| Molecular Formula | C19H30O3 |
| Molecular Weight | 306.44 g/mol |
| SMILES |
C[C@@H]1C[C@H]2[C@@H]3CC[C@](O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]4(C)C=CC(=O)C[C@@H]14 |
| InChIKey |
QSLJIVKCVHQPLV-PEMPUTJUSA-N |
| Melting Point | 235-238 °C |
| Solubility | Slightly soluble in water; soluble in acetone, chloroform |
| Half-life | 9 hours (oral) |
Pharmacological Profile
| Anabolic Rating | 322 |
|---|---|
| Androgenic Rating | 24 |
| Aromatisation | None |
| Hepatotoxicity | Low |
| Detection Window | 3 weeks |
Known trade names
- Anavar
- Oxandrin
- Lonavar
Products containing Oxandrolone
Cycles using this compound
External references
Data sourced from peer-reviewed pharmacological literature and authoritative chemical databases (PubChem, DrugBank, ChEBI). Provided for identification and research reference only — not medical advice.