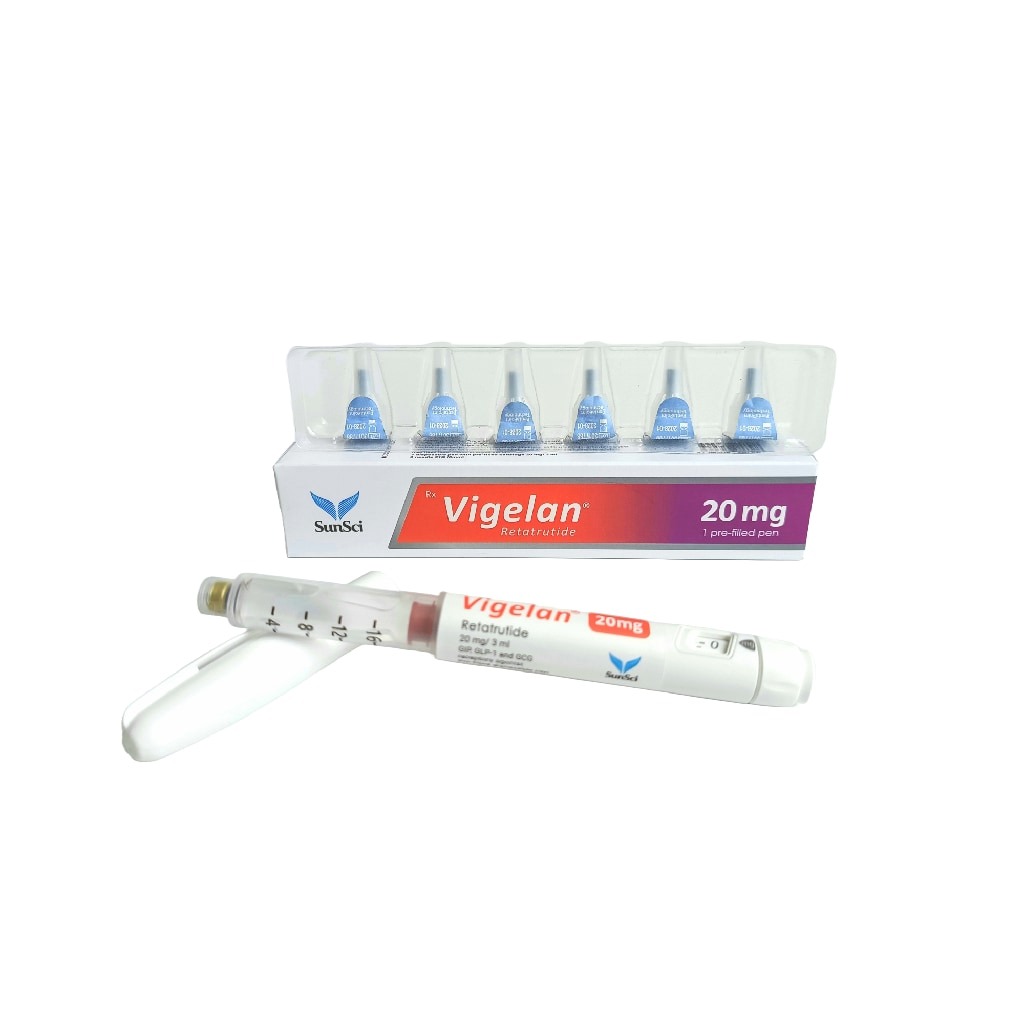
Description
Vigelan (Retatrutide) 20 mg SunSci Pharmaceuticals — retatrutide from SunSci Pharmaceuticals at 20 mg. Triple agonist (GLP-1 + GIP + glucagon receptors). Largest documented weight-loss effect of any injectable in published trials; still in late-phase research.
Key Benefits
- Triple agonist — GIP + GLP-1 + glucagon receptors
- Phase-2 data: ~24% body-weight reduction at 48 weeks
- Glucagon component drives thermogenesis and hepatic fat oxidation
- Currently investigational — not yet FDA-approved
- Weekly SubQ dosing with titration
- Each unit dosed at 20 mg — see Recommended Dosage below for protocol-specific intake
How It Works
First-in-class triple agonist combining GLP-1 and GIP incretin effects with glucagon-receptor activation. Glucagon component drives thermogenesis and hepatic lipolysis — mechanism absent from semaglutide/tirzepatide. Investigational; not approved as of 2024.
Pharmacokinetics
Plasma half-life approximately 6 days. Weekly SubQ dosing. Phase-2 data show dose-proportional exposure and stable steady-state by week 4.
Potential Side Effects
GI side-effects higher than semaglutide/tirzepatide during titration. Heart-rate elevation modest. Muscle-mass loss requires training offset. Long-term safety profile still being established — use with awareness of investigational status.
Cycle & Stacking Guide
Investigational protocol. Research-grade use only. Resistance training and high-protein diet essential for lean-mass preservation. Follow phase-2 trial titration schedule closely.
Manufacturer Notes
SunSci Pharmaceuticals produces semaglutide pens and growth hormone products. Cold-chain shipping for all biological products.
Storage & Handling
Store at recommended temperature (15–25°C; peptides and HGH at 2–8°C after reconstitution). Protect from light and moisture. Keep out of reach of children. For research and educational purposes only.


Reviews
There are no reviews yet.