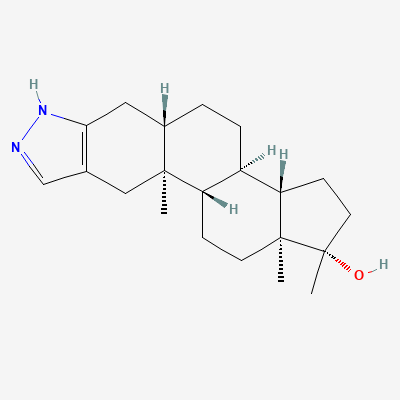
Stanozolol
-
CAS
10418-03-8 - Formula C21H32N2O
- Half-life 9 hours (oral) / 24 hours (IM)
- Detection 2 months
Clinical Notes
Heterocyclic DHT derivative — pyrazole fused to the A-ring, blocking 3alpha-HSD metabolism. Crushes SHBG in a dose-responsive manner (documented 40–50% reduction at 50 mg/day oral x 4 weeks), which is the proximate mechanism for its perceived amplification effect on co-administered androgens: bound testosterone is released, free-testosterone fraction rises. The compound itself has modest direct AR activity. Oral form is C17alpha-alkylated with predictable hepatotoxicity. Connective-tissue profile is the clinical liability: tendinopathy and ligament rupture frequency is elevated on stanozolol protocols. Joint lubrication complaint is universal above 40 mg/day and resolves on cessation. Lipid degradation is among the steepest in the oral class; HDL reductions of 50–60% at therapeutic doses are reproducible.
Chemical Identity
| CAS Number |
10418-03-8 |
|---|---|
| IUPAC Name | (1S,3aS,3bR,5aS,10aS,10bS,12aS)-1,10a,12a-trimethyl-2,3,3a,3b,4,5,5a,6,10b,11,12,12a-dodecahydro-1H-cyclopenta[5,6]naphtho[1,2-f]indazol-1-ol |
| Molecular Formula | C21H32N2O |
| Molecular Weight | 328.49 g/mol |
| SMILES |
C[C@]1(O)CC[C@@H]2[C@H]3CCC4=CC5=CC=N[NH]5C[C@]4(C)[C@@H]3CC[C@]12C |
| InChIKey |
HCNHNBLBDSXWPN-UHFFFAOYSA-N |
| Melting Point | 229-242 °C |
| Solubility | Practically insoluble in water; soluble in dimethylformamide |
| Half-life | 9 hours (oral) / 24 hours (IM) |
Pharmacological Profile
| Anabolic Rating | 320 |
|---|---|
| Androgenic Rating | 30 |
| Aromatisation | None |
| Hepatotoxicity | High |
| Detection Window | 2 months |
Known trade names
- Winstrol
- Stromba
- Stanover
Products containing Stanozolol
External references
Data sourced from peer-reviewed pharmacological literature and authoritative chemical databases (PubChem, DrugBank, ChEBI). Provided for identification and research reference only — not medical advice.