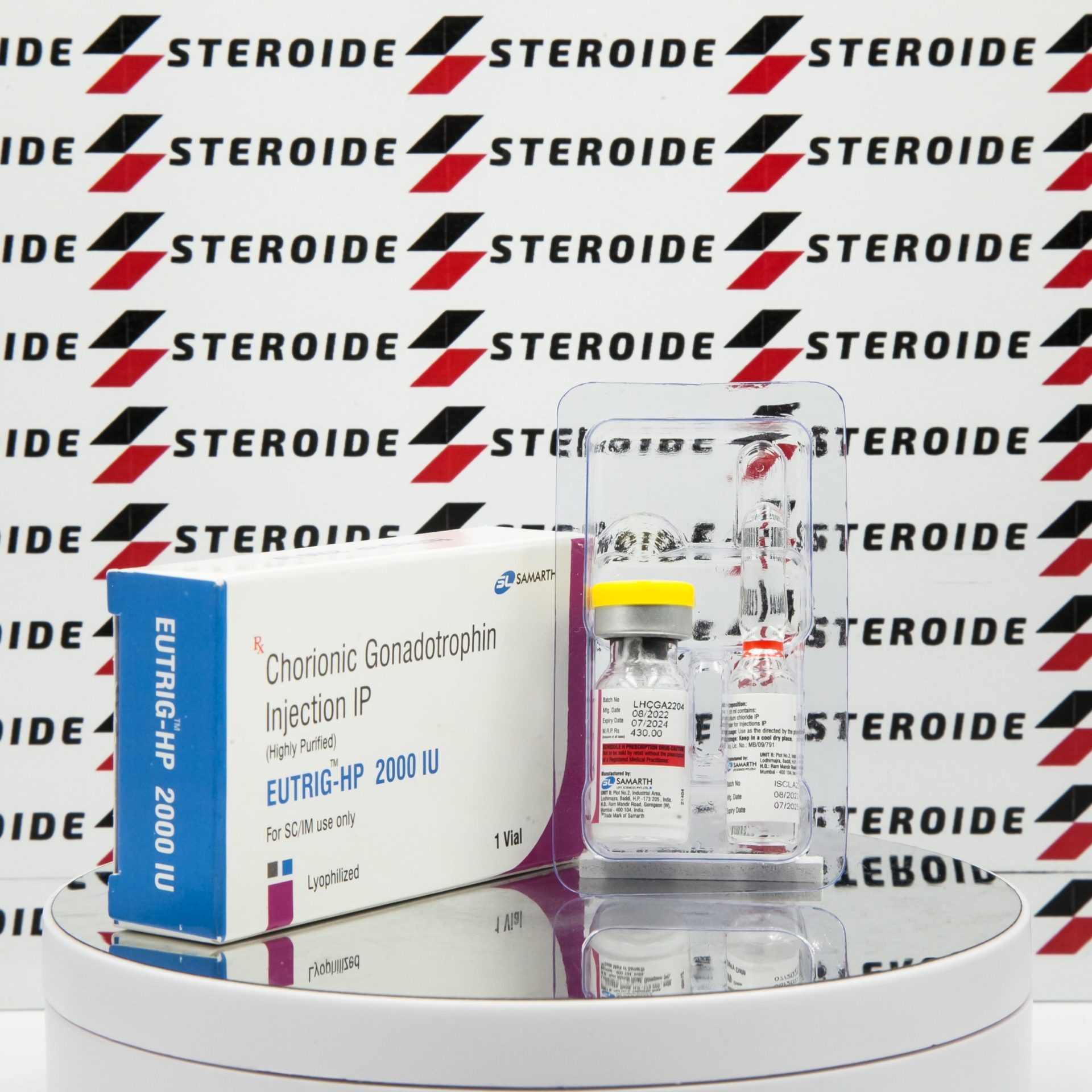
Description
Chorionic Gonadotropin Injection IP EUTRIG-HP 2000 IU Samarth — human chorionic gonadotropin from Samarth at 2000 iu. LH-mimetic: directly stimulates leydig-cell testosterone production and spermatogenesis. On-cycle testicular maintenance or pre-PCT priming.
Key Benefits
- Human chorionic gonadotropin — mimics LH, stimulates testicular testosterone
- Prevents testicular atrophy on long cycles
- Used on-cycle (not during PCT — can desensitise receptors)
- Reconstituted with bacteriostatic water; SubQ or IM injection
- Standard on-cycle 250–500 IU 2× weekly
- Each unit dosed at 2000 IU — see Recommended Dosage below for protocol-specific intake
Recommended Dosage
Research dosing: on-cycle 250–500 IU 2× weekly SubQ to prevent testicular atrophy. Pre-PCT blast 1000 IU 2× weekly for 2 weeks. Not used during PCT proper — chronic HCG desensitises Leydig cells to LH.
How It Works
Human chorionic gonadotropin, a glycoprotein hormone with LH-like activity. Binds the LH receptor on testicular Leydig cells, stimulating testosterone production directly — bypasses pituitary suppression.
Pharmacokinetics
Plasma half-life approximately 33 hours. Twice-weekly SubQ dosing produces stable effect. Reconstituted vial stable in fridge for 30 days.
Potential Side Effects
Excessive dose raises E2 (testicular estrogen output) — pair with AI. Water retention possible. Chronic high-dose use desensitises LH receptors, hampering natural recovery.
Cycle & Stacking Guide
On-cycle protocol: 500 IU 2× weekly throughout the cycle to maintain testicular size and function. Stop 2 weeks before SERM-based PCT begins. Alternative: pre-PCT blast approach — skip during cycle, then 1000 IU × 2/week for 2 weeks before starting SERMs.
Manufacturer Notes
Samarth is an independent producer in the research-compound market. Before first use, request the batch-specific Certificate of Analysis and confirm the vial’s tamper-evident seal is intact.
Storage & Handling
Pre-reconstitution: store at 2–8 °C, protected from freezing and light. Post-reconstitution: use within 14–21 days, maintained at 2–8 °C throughout. Never freeze reconstituted solution — freezing irreversibly denatures the protein. Transport in an insulated cold-pack for trips. Keep out of reach of children. For research and educational purposes only.

Reviews
There are no reviews yet.